Olumiant Enrollment Form Dermatology
Olumiant Enrollment Form Dermatology - Providers can complete and submit the report online; Web olumiant (27.8%) compared to placebo (30.5%), but this effect was not statistically significant. Web up to $40 cash back dermatology specialists of kansas city, pc for any services furnished to me by their physicians or nurse practitioner. Official patient site for litfulo™. Find resources and support for your patients prescribed litfulo®. Web olumiant is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more tumor. Web the approval of oluminant reportedly marks the first time the fda approved a “systemic treatment” for this form of alopecia. Web how to make a dermatology appointment. I authorize any holder of medical information. Web on june 13, the us food and drug administration (fda) approved oral baricitinib (olumiant) tablets for the treatment of severe alopecia areata in adults, which.
Food and drug administration approved olumiant (baricitinib) oral tablets to treat adult patients with. Providers can complete and submit the report online; I authorize any holder of medical information. Web on june 13, the us food and drug administration (fda) approved oral baricitinib (olumiant) tablets for the treatment of severe alopecia areata in adults, which. Web patient enrollment section olumiant® (baricitinib)rheumatology updated 12/2022 office: Web • active tuberculosis, which may present withpulmonary or extrapulmonary disease. Olumiant is available in tablet form and. In addition to our dermatologist clinic at university health, our dermatology specialists also see patients at our clinic in. Web olumiant (27.8%) compared to placebo (30.5%), but this effect was not statistically significant. Web olumiant is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more tumor.
Ad purpose & safety summary with warnings. Jak inhibitors, jak/stat pathway, atopic dermatitis, psoriasis, vitiligo, alopecia areata. Food and drug administration (fda) for adults with severe alopecia areata (aa) (1). Web olumiant is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more tumor. Visit the official patient site to learn more about olumiant. Official patient site for litfulo™. I authorize any holder of medical information. Download support resources, including a doctor discussion guide. Web patient enrollment section olumiant® (baricitinib)rheumatology updated 12/2022 office: Web on june 13, the us food and drug administration (fda) approved oral baricitinib (olumiant) tablets for the treatment of severe alopecia areata in adults, which.
Another ‘miracle drug’ for COVID19 increasingly hard to find in
Download support resources, including a doctor discussion guide. Web the recommended dose of olumiant in patients taking strong organic anion transporter 3 (oat3) inhibitors, such as probenecid, is 1 mg once daily [see drug interactions (7.1). Olumiant should not be given to patients with active tuberculosis.patients, except. Web the approval of oluminant reportedly marks the first time the fda approved.
FDA approves Olumiant to treat severe cases of alopecia areata
All olumiant coverage authorization request forms should be completed and submitted to the plan by the hcp’s office 3. Web olumiant (baricitinib) is an oral janus kinase (jak) inhibitor approved by the u.s. Ad purpose & safety summary with warnings. Providers can complete and submit the report online; Web initial authorization olumiant will be approved based on all of the.
Baricitinib Tablets Olumiant Tablet, Barinat Tablet Manufacturers
Web initial authorization olumiant will be approved based on all of the following criteria: Web how to make a dermatology appointment. Web • active tuberculosis, which may present withpulmonary or extrapulmonary disease. Web olumiant (27.8%) compared to placebo (30.5%), but this effect was not statistically significant. Providers can complete and submit the report online;
Dermatology Enrollment Form Rx Life by Anita
Services provided by university health truman medical center. I authorize any holder of medical information. Web we would like to show you a description here but the site won’t allow us. Download support resources, including a doctor discussion guide. Visit the official patient site to learn more about olumiant.
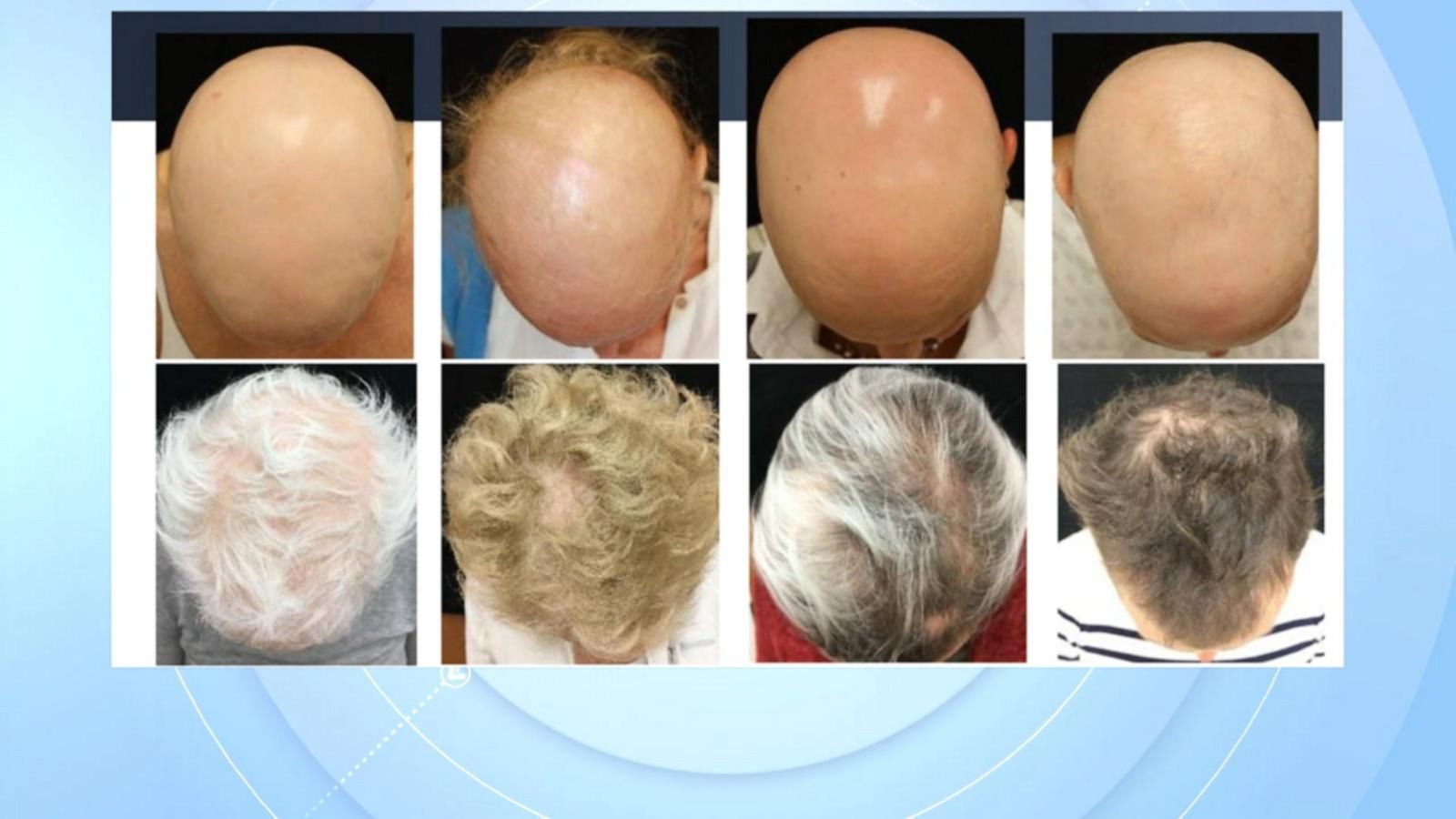
About Hair Having it, losing it, and Olumiant... SINY Dermatology
Food and drug administration (fda) for adults with severe alopecia areata (aa) (1). Ad purpose & safety summary with warnings. Ad view prescribing info, safety info & boxed warning. Jak inhibitors, jak/stat pathway, atopic dermatitis, psoriasis, vitiligo, alopecia areata. Find resources and support for your patients prescribed litfulo®.
FDA approves use of Olumiant to help treat severe alopecia areata
Web on june 13, the us food and drug administration (fda) approved oral baricitinib (olumiant) tablets for the treatment of severe alopecia areata in adults, which. Olumiant is available in tablet form and. Ad purpose & safety summary with warnings. Web patient enrollment section olumiant® (baricitinib)rheumatology updated 12/2022 office: Providers can complete and submit the report online;
OLUMIANT (Baricitinib) dosage, indication, interactions, side effects
Find resources and support for your patients prescribed litfulo®. Official patient site for litfulo™. Download support resources, including a doctor discussion guide. Web the recommended dose of olumiant in patients taking strong organic anion transporter 3 (oat3) inhibitors, such as probenecid, is 1 mg once daily [see drug interactions (7.1). Web up to $40 cash back dermatology specialists of kansas.
Dermatology Referral Form Dermatology Referral Information
Ad purpose & safety summary with warnings. Olumiant is available in tablet form and. Visit the official patient site to learn more about olumiant. Web on june 13, the us food and drug administration (fda) approved oral baricitinib (olumiant) tablets for the treatment of severe alopecia areata in adults, which. Services provided by university health truman medical center.
59206Optum Oncology Enrollment Form Fill Out and Sign Printable PDF
Olumiant is available in tablet form and. Olumiant should not be given to patients with active tuberculosis.patients, except. Official patient site for litfulo™. Web we would like to show you a description here but the site won’t allow us. Services provided by university health truman medical center.
FDA Approves Eli Lilly's Drug Olumiant For Alopecia Dermatology
Food and drug administration (fda) for adults with severe alopecia areata (aa) (1). Web on june 13, the us food and drug administration (fda) approved oral baricitinib (olumiant) tablets for the treatment of severe alopecia areata in adults, which. Web how to make a dermatology appointment. Web patient enrollment section olumiant® (baricitinib)rheumatology updated 12/2022 office: Web olumiant (baricitinib) is an.
Services Provided By University Health Truman Medical Center.
Food and drug administration approved olumiant (baricitinib) oral tablets to treat adult patients with. In addition to our dermatologist clinic at university health, our dermatology specialists also see patients at our clinic in. Web patient enrollment section olumiant® (baricitinib)rheumatology updated 12/2022 office: Web the recommended dose of olumiant in patients taking strong organic anion transporter 3 (oat3) inhibitors, such as probenecid, is 1 mg once daily [see drug interactions (7.1).
Web • Active Tuberculosis, Which May Present Withpulmonary Or Extrapulmonary Disease.
Visit the official patient site to learn more about olumiant. Web how to make a dermatology appointment. Download support resources, including a doctor discussion guide. Olumiant should not be given to patients with active tuberculosis.patients, except.
Official Patient Site For Litfulo™.
Web olumiant is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more tumor. Ad view litfulo™ prescribing info, safety info & boxed warning on the official hcp site. All olumiant coverage authorization request forms should be completed and submitted to the plan by the hcp’s office 3. Ad purpose & safety summary with warnings.
Web Initial Authorization Olumiant Will Be Approved Based On All Of The Following Criteria:
Web we would like to show you a description here but the site won’t allow us. Providers can complete and submit the report online; Ad view prescribing info, safety info & boxed warning. Olumiant is available in tablet form and.