What Elements Form Negative Ions
What Elements Form Negative Ions - Web the elements that will form negative ions are those that have more electrons than protons in their nucleus. Positively charged ions are called cations; Web nitrogen’s position in the periodic table (group 15) reveals that it is a nonmetal. Metals never form negative ions. Web an ion is an atom or molecule that has a different number of electrons than protons, so it has a charge. All the halogens gain a single electron to fill their valence energy level. Web what elements form negative ions? Those elements in groups 5, 6 and 7 which form simple negative ions all have noble gas structures. Nonmetals form negative ions (anions). 4 comments ( 106 votes) upvote downvote flag
Web negative ions, known as anions, form when an atom gains electrons and now has more electrons than protons, indicating that the number of protons and electrons is not. Positively charged ions are called cations; Nonmetals elements that easily gain electrons to form negative ions? Web compounds formed from positive and negative ions are ionic compounds. Individual atoms can gain or lose electrons. Web an ion that has more electrons than protons, giving it a net negative charge, is named an anion, and a minus indication anion (−) indicates the negative charge. All the halogens gain a single electron to fill their valence energy level. Web an ion is an atom or molecule that has a different number of electrons than protons, so it has a charge. Clearly the parent atoms (or molecules) are. Negatively charged ions are called.
Those elements in groups 5, 6 and 7 which form simple negative ions all have noble gas structures. Web anions are the negative ions formed from the gain of one or more electrons. Web nitrogen’s position in the periodic table (group 15) reveals that it is a nonmetal. The parent atoms are oxygenating species: Clearly the parent atoms (or molecules) are. Web negative ions are tidier! Negatively charged ions are called. The ions formed are negative, because they have more electrons than protons. Web when these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. Web the elements that will form negative ions are those that have more electrons than protons in their nucleus.
The Chemistry of Ion Exchange WCP Online
Tend to form negative ions, i.e. If elements are not aiming for noble. These include the alkali metals, such as sodium and. When they do, they become monatomic ions. Web an ion that has more electrons than protons, giving it a net negative charge, is named an anion, and a minus indication anion (−) indicates the negative charge.
SOLVEDAn element forms a negative ion when ionized. On what side of
4 comments ( 106 votes) upvote downvote flag Web negative ions, known as anions, form when an atom gains electrons and now has more electrons than protons, indicating that the number of protons and electrons is not. Clearly the parent atoms (or molecules) are. And all of them form an. Web what elements form negative ions?
Periodic Table Ionic Charges Pdf Awesome Home
Web an ion that has more electrons than protons, giving it a net negative charge, is named an anion, and a minus indication anion (−) indicates the negative charge. Web moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the.
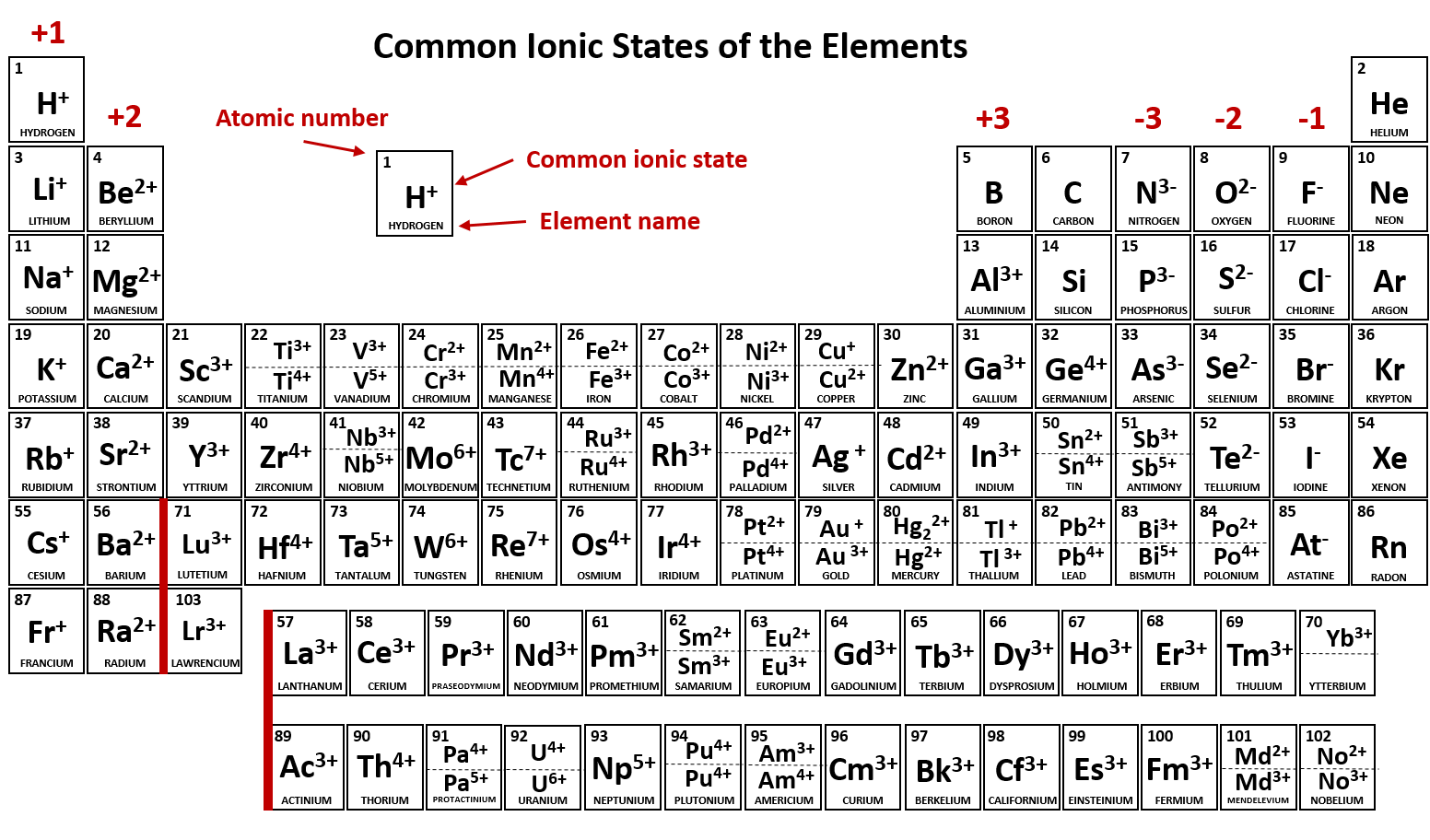
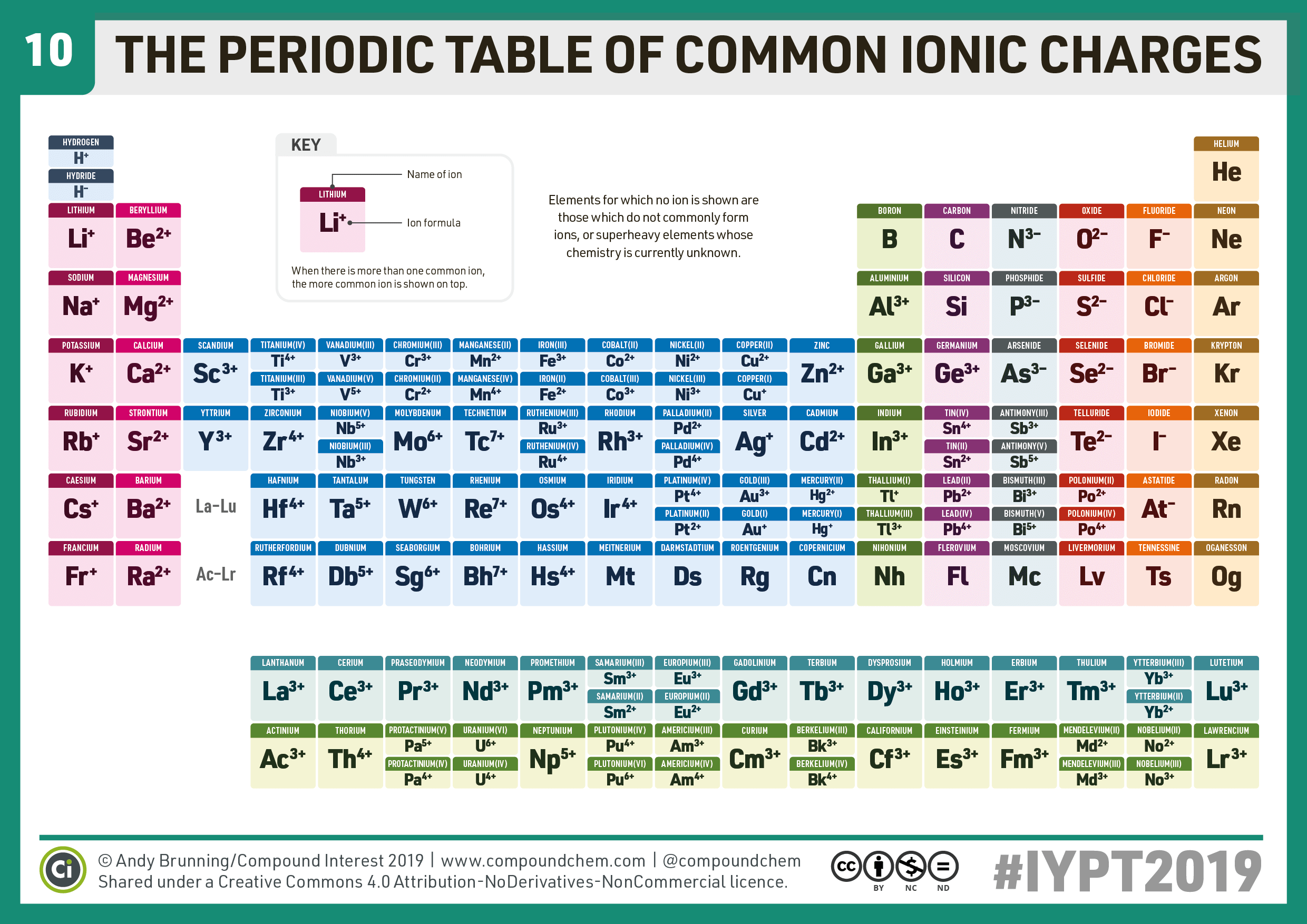
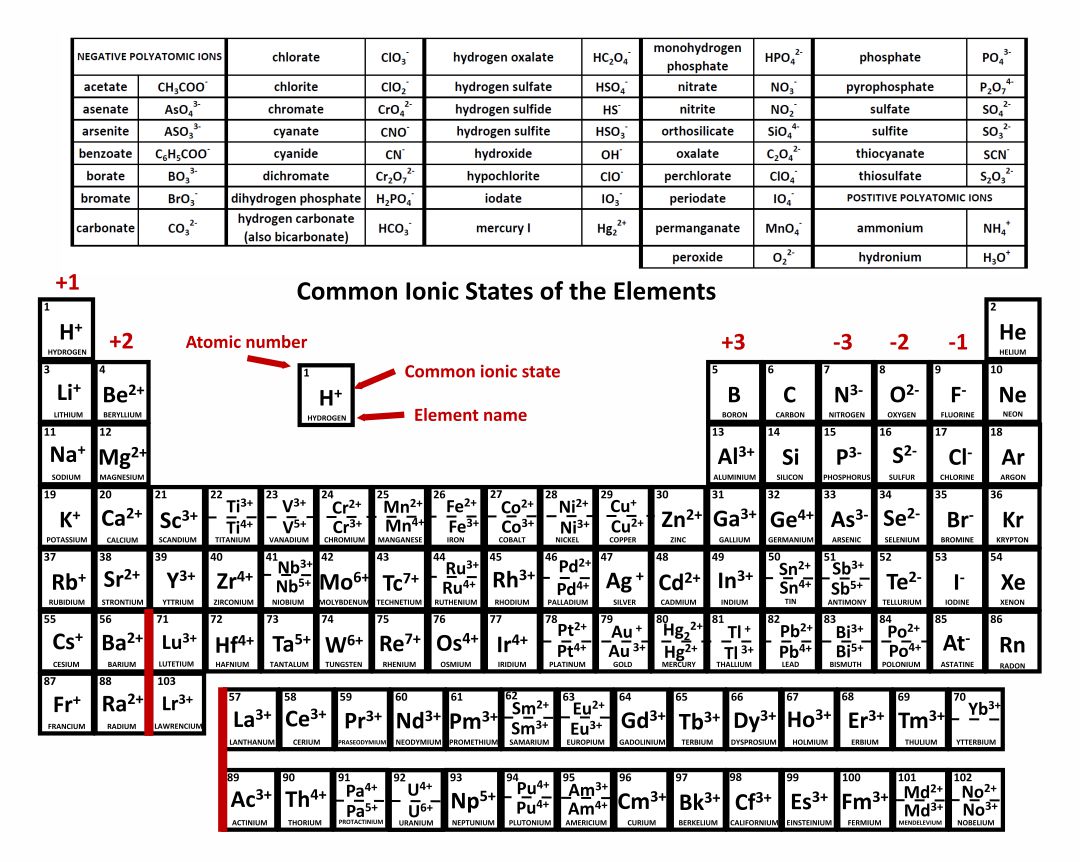
10 Periodic Table of Common Ions Compound Interest
Positively charged ions are called cations; Web atoms gain electrons in their outer shell when they form negative ions, called anions. Web an ion is an atom or molecule that has a different number of electrons than protons, so it has a charge. The parent atoms are oxygenating species: Web anions are the negative ions formed from the gain of.
Ions
The ions formed are negative, because they have more electrons than protons. Web the halogens (viia elements) all have seven valence electrons. Web what elements form negative ions? All the halogens gain a single electron to fill their valence energy level. Web the elements that will form negative ions are those that have more electrons than protons in their nucleus.
CH150 Chapter 3 Ions and Ionic Compounds Chemistry
If elements are not aiming for noble. When nonmetal atoms gain electrons, they often do so until their outermost principal. Negatively charged ions are called. Web the elements that will form negative ions are those that have more electrons than protons in their nucleus. Web what elements form negative ions?
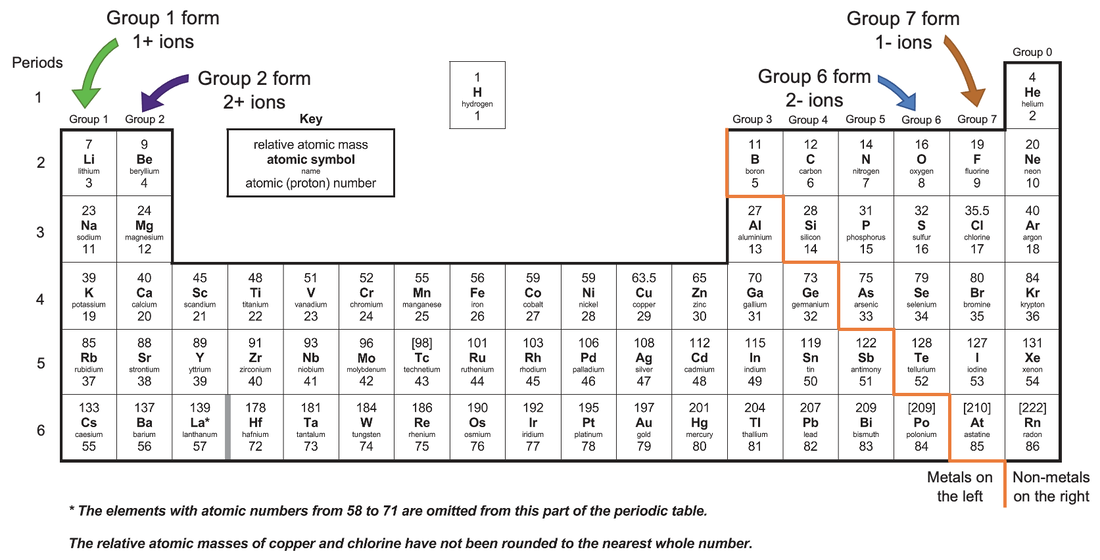
C2 B) Ions from the Periodic Table AQA Combined Science Trilogy Elevise
Ion, any atom or group of atoms that bears one or more positive or negative electrical charges. And all of them form an. Web atoms gain electrons in their outer shell when they form negative ions, called anions. Web nitrogen’s position in the periodic table (group 15) reveals that it is a nonmetal. Nonmetals form negative ions (anions).
Electron Affinity of The Elements
Web when these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. If elements are not aiming for noble. Web nitrogen’s position in the periodic table (group 15) reveals that it is a nonmetal. Clearly the parent atoms (or molecules) are. Web compounds formed from positive and negative ions are ionic compounds.
10 Best Printable Periodic Table Of Ions
Web an ion is an atom or molecule that has a different number of electrons than protons, so it has a charge. And all of them form an. Web the halogens (viia elements) all have seven valence electrons. The parent atoms are oxygenating species: A nitrogen atom must gain three electrons to have the.
3.2 Ions The Basics of General, Organic, and Biological Chemistry
All the halogens gain a single electron to fill their valence energy level. Tend to form negative ions, i.e. Web the elements that will form negative ions are those that have more electrons than protons in their nucleus. Metals never form negative ions. Clearly the parent atoms (or molecules) are.
Produce Negative Ions, Such As Oxygen.
Web the halogens (viia elements) all have seven valence electrons. Web anions are the negative ions formed from the gain of one or more electrons. Web in this way, nitrogen, oxygen, fluorine, chlorine, etc., are formed. Web the elements that will form negative ions are those that have more electrons than protons in their nucleus.
N 3−, O2−, F −, And Cl−;
Positively charged ions are called cations; When they do, they become monatomic ions. Web an ion that has more electrons than protons, giving it a net negative charge, is named an anion, and a minus indication anion (−) indicates the negative charge. If elements are not aiming for noble.
Web Atoms Gain Electrons In Their Outer Shell When They Form Negative Ions, Called Anions.
Individual atoms can gain or lose electrons. Tend to form negative ions, i.e. Web what elements form negative ions? Clearly the parent atoms (or molecules) are.
Web Moving From The Far Right To The Left On The Periodic Table, Elements Often Form Anions With A Negative Charge Equal To The Number Of Groups Moved Left From The Noble Gases.
Web compounds formed from positive and negative ions are ionic compounds. Web and thus nitrogen, oxygen, fluorine, chlorine, etc. Metals never form negative ions. Web when these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons.